Hard Gelatin Capsule
Manufacture of hard gelatin capsules includes the following steps:
Manufacture of capsule shells
Extraction of gelatin: Gelatin is a heterogeneous product derived by irreversible hydrolytic extraction of treated collagen obtained from animal bones and skin (Fig. 5.2). The properties of gelatin that are to be evaluated are as follows:
- Bloom Strength: It is defined as assessment of cohesive strength of cross-linking taking place in between the molecules of gelatin. It is also known as gel strength of gelatin and depends on the molecular weight of gelatin. The harder the gelatin higher the bloom strength. The weight required to move a standard plunger in to standard
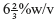 gelatin solution determines the bloom strength of gelatin. Bloom strength must be between 150 and 250 bloom grams.
gelatin solution determines the bloom strength of gelatin. Bloom strength must be between 150 and 250 bloom grams. - Viscosity: Viscosity of gelatin is required to control the thickness of the film, which is measured by standard
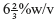 gelatin solution maintained at 60 °C. The range of viscosity should be between 30 and 60 millipoise.
gelatin solution maintained at 60 °C. The range of viscosity should be between 30 and 60 millipoise. - Iron content: Iron is present in both raw gelatin and water used for manufacturing gelatin capsule shells. Excess amount of iron can affect Food drug and cosmetics (FDC) certified dyes and may react with other organic compounds. Iron is used in a concentration not more than 15 ppm.
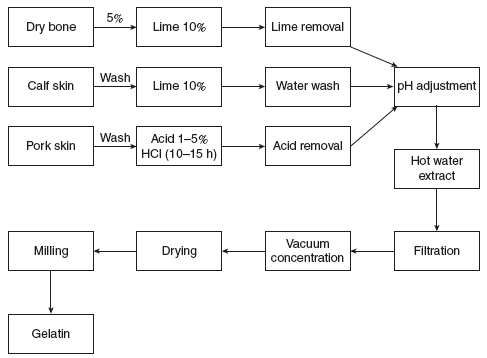 Figure 5.2 Schematic Representation for Extraction of Gelatin
Figure 5.2 Schematic Representation for Extraction of Gelatin
Two types of gelatin used are as follows:
- Type A gelatin is derived from acid treated precursor and exhibits an isoelectric point in the region of pH 9.
- Type B gelatin is derived from alkali treated precursor and has an isoelectric point of pH 4.7.
Capsules can be made from either type of gelatin but usually a mixture of both is used. Bone gelatin produces a tough film but it becomes hazy and brittle. However, the pork skin gelatin contributes the plasticity, clarity and elasticity producing a transparent film. Blends of bone and pork skin gelatin of relatively high gel strength are normally used for hard capsule shell production.
Preparation of gelatin solution
- Gelatin—Gelatin extracted of Type A and Type B are used.
- Plasticizer— normally used are glycerin or sorbitol or in combinations. The ratio of gelatin and plasticizer is (0.4:1).
- Preservatives—usually mixture of 0.2% w/v of methyl paraben and propyl paraben are used. Others like sodium sulfite or sodium metabisulpfite can also be used.
- Coloring agents—Various FDC approved dyes and pigments are used.
- Opacifying agents—Titanium dioxide (0.3%) is generally used to make the shell opaque, which is specially used to provide protection against light.
- Sugar—included in the formulation up to 5%, which acts as a sweetening agent and also increases the viscosity of the gelatin solution.
- Demineralized water—used as a vehicle for the gelatin solution preparation.
Processing of capsule shells
- Dipping: The required pairs of stainless steel mould pegs or pins (long length with lesser diameter for body and short length with larger diameter for cap) are dipped in gelatin solution for a period of 12 s, which helps in achieving proper length and thickness of shells.
- Spinning: After dipping, the pins are rotated for uniform distribution of gelatin solution and withdrawn with a blast of cold air.
- Drying: The gelatin coated pins are introduced into the drying chamber and dried at specified temperature and humidity.
- Stripping: After drying, the cap and body portions are stripped off from the pins by using jaws made of bronze.
- Trimming: The body and cap portions are trimmed to required sizes. The trimmed cap and body are joined and capsule shells are sent to sorting section for inspection.
Formulation of the blend
- Drug: Medicament may be single or multicomponent.
- Bulking agent/Diluents: If the dose of the drug is very less and to increase the bulk volume for filling of the capsules as per the size of the capsule shell selected, bulking agent is used. For example, lactose, starch, sucrose, and mannitol.
- Disintegrating agent: To enhance the rate of disintegration of capsules, especially for water insoluble drugs are used. For example, starch, sodium CMC, PVP and sodium starch glycolate.
- Glidant and lubricant: For the easy flow of the powder blend from the hopper in to the body of capsule shell and also for easy ejection of the filled capsule from the capsule filling machine the above ingredients are added. For example, purified talc and magnesium stearate.
Filling, sealing and packing of capsules
Capsules are manufactured as depicted in Figure 5.3.
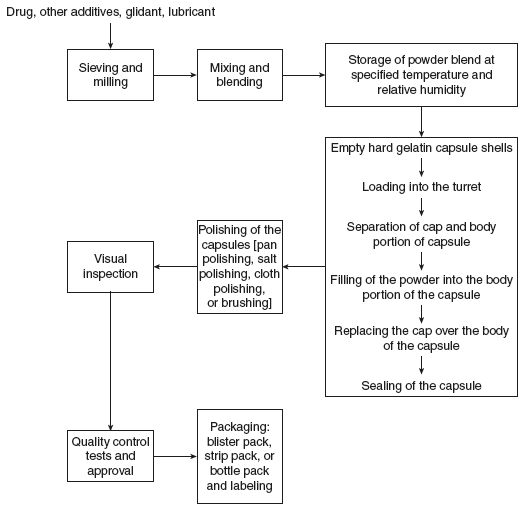
Figure 5.3 Process Layout of Capsule Manufacture
Leave a Reply